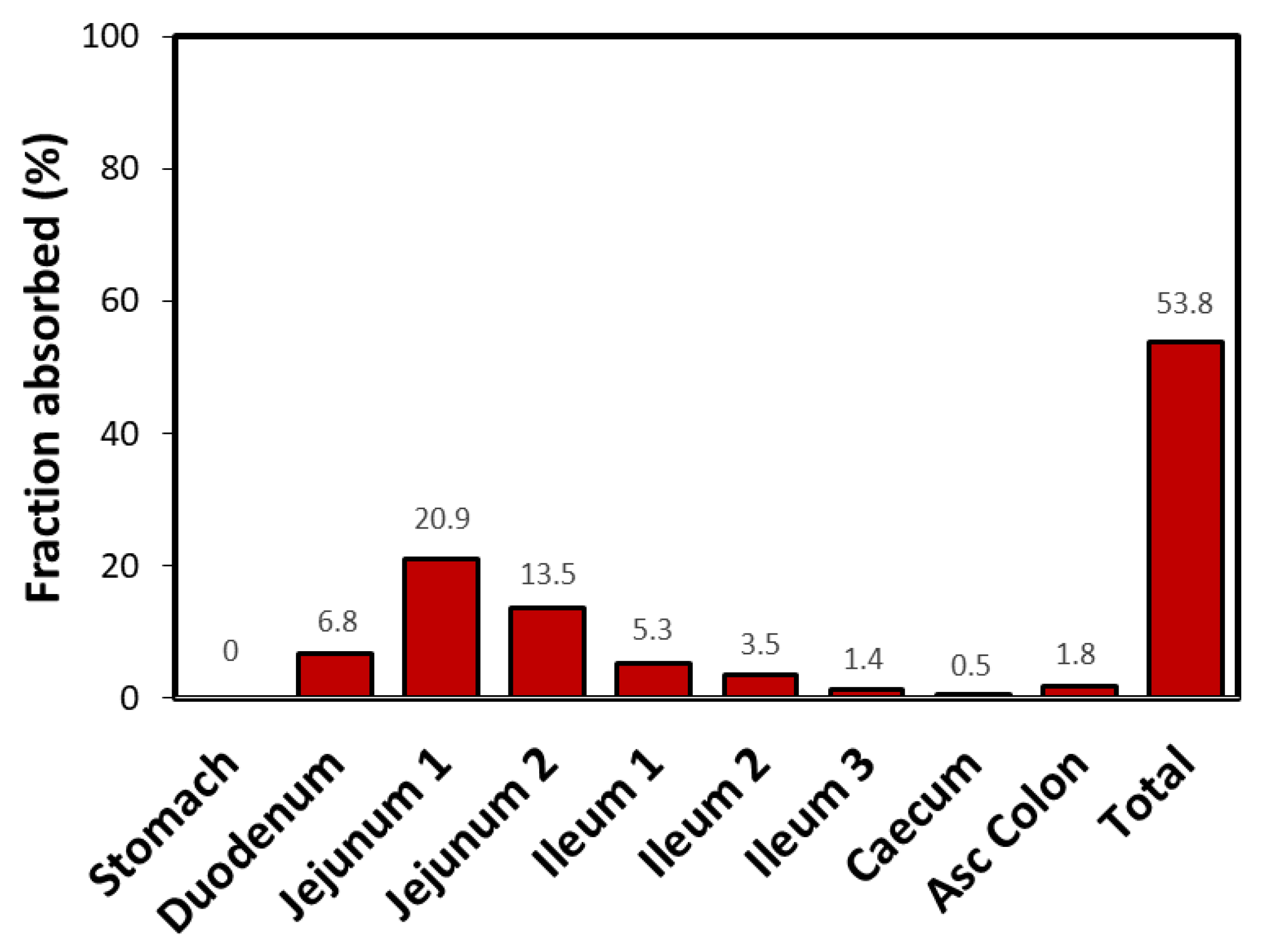
There were no clear associations between a drug’s F in dogs and humans or between the canine value of F and either its human BCS classification, its LogP value, or the canine Do estimate. Whether using 35 or 6 mL to determine Do, the canine solubility classification remained unchanged for all but seven compounds. These same criteria were applied for defining highly soluble and highly permeable in dogs. In addition, in humans, a Do value greater than 1.0 is used to define a compound as highly soluble and a LogP value greater than 1.72 as high permeability. In this analysis, the estimated volume of a water flush administered to fasted dogs (6 mL) and a volume of 250 mL scaled to a Beagle dog (35 mL) were examined. However, the appropriate volume for classifying drug solubility in dogs has not been established.

In humans, solubility is determined in 250 mL of fluid.

The drugs were also evaluated for any potential association between solubility (calculated from the dose number, Do) or lipophilicity (LogP) and F in dogs. Therefore, we examined 50 drugs for which absolute bioavailability ( F) was available both in dogs and humans.

To date, the applicability of existing human BCS criteria has not been evaluated in dogs, thereby limiting its use in canine drug development. When used in conjunction with in vitro dissolution tests, the BCS can support the prediction of in vivo product performance and the development of mechanistic models that support formulation assessments through the generation of “what if” scenarios. For example, some drugs or compounds may be deemed a schedule I drug or may be listed in a different schedule in a state's specific controlled substance act, which may differ from the federal controlled substance act.The Biopharmaceutical Classification System (BCS) has been a prognostic tool for assessing the potential effects of formulation on the human drug oral bioavailability. There may be variations in CSA schedules between individual states and federal law. The Controlled Substances Act (CSA) schedule information displayed above applies to drugs or substances regulated under federal law. Bath Salts (3,4-methylenedioxypyrovalerone or MDPV).Synthetic marijuana and analogs (Spice, K2).Ecstasy (MDMA or 3,4-Methylenedioxymethamphetamine).GHB (gamma-hydroxybutyric acid) - except formulations in an FDA-approved drug product sodium oxybate ( Xyrem) are Schedule III.MDMA (3,4-methylenedioxymethamphetamine or “ecstasy”).Read More: Marijuana: Effects, Medical Uses & Legalization Drugs or Substances listed in DEA Schedule I may include: states have legalized marijuana for personal, recreational use or for medical use. NOTE: Tetrahydrocannabinol (THC, marijuana) is still considered a Schedule 1 drug by the DEA, even though some U.S. A controlled substance analogue (for example, a "designer drug") is a substance which is structurally or pharmacologically similar to a Schedule I or Schedule II substance, specifically used for human consumption, and is not an approved medication in the United States. Learn More: Commonly Abused Drugs and SubstancesĪ substance does not need to be listed as a controlled substance by the DEA to be treated as a Schedule I substance for criminal prosecution. It has a lack of accepted safety for use under medical supervision.Īccording to federal law, no prescriptions may be written for Schedule I substances, and they are not readily available for clinical use.The drug or other substance has no currently accepted medical treatment use in the U.S.The drug or other substance has a high potential for abuse.Schedule I drugs are those that have the following characteristic according to the United States Drug Enforcement Agency (DEA): Medically reviewed by Leigh Ann Anderson, PharmD.
